Vandria is a Swiss biotechnology company investigating the potential of mitochondrial therapeutics to combat age-related and chronic diseases.
Their primary strategy involves developing small-molecule mitophagy inducers, which are compounds that promote the selective removal and replacement of damaged mitochondria within cells. By enhancing mitophagy, Vandria aims to rejuvenate cellular function, thereby addressing conditions such as neurodegenerative disorders and muscle diseases.
Feel welcome to share your own thoughts on this company in the comment section below as well. I will be happy to discuss and learn more about how you see their potential in this field.
The Solution
Lead Program: VNA-318
VNA-318 is Vandria’s flagship compound, designed to treat central nervous system (CNS) disorders such as mild cognitive impairment, Alzheimer’s disease, and Parkinson’s disease. This orally available, brain-penetrant small molecule operates through a novel target associated with several human diseases, including Alzheimer’s.
VNA-318 exhibits a dual mode of action:
- Immediate Effects:
Enhances memory, learning, and cognitive function. - Long-Term Benefits:
Provides disease-modifying effects, including reduced neuroinflammation, decreased toxic protein aggregation, and improved mitochondrial function.
Preclinical studies have demonstrated VNA-318’s efficacy in models of neurodegenerative diseases, showing both acute cognitive improvements and long-term neuroprotective benefits. Toxicity studies have indicated a wide safety margin for the compound. A composition of matter patent covering VNA-318 and related compounds has been issued by the U.S. Patent Office.
As of December 2024, Vandria has initiated its first-in-human Phase 1 clinical trial for VNA-318, dosing the initial subjects. This randomized, double-blind study is designed to assess safety, tolerability, pharmacokinetics, and pharmacodynamics in healthy male participants. Results from this combined single and multiple ascending dose trial are anticipated in the summer of 2025.
Pipeline
Beyond VNA-318, Vandria is developing a pipeline of mitophagy-inducing small molecules, each tailored for specific pharmacokinetic properties to target different tissues and address various unmet medical needs:
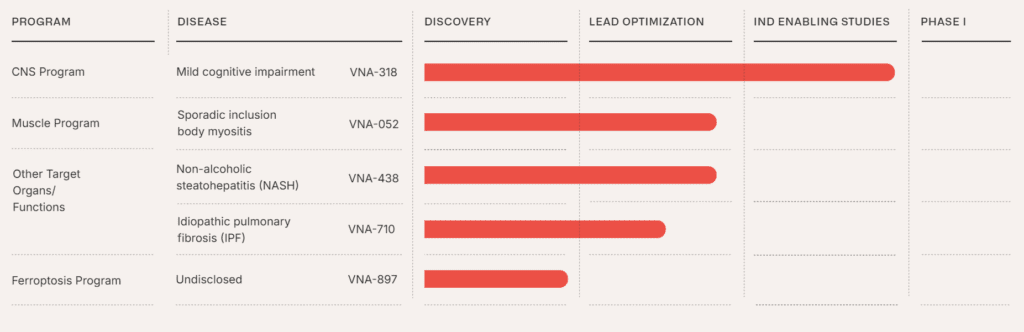
- VNA-052: Aimed at treating muscle diseases, particularly sporadic inclusion body myositis.
- VNA-438: Developed for non-alcoholic steatohepatitis (NASH), a chronic liver disease.
- VNA-710: Intended for idiopathic pulmonary fibrosis (IPF), a severe lung condition.
- VNA-897: Focused on ferroptosis, a form of cell death implicated in neurodegenerative diseases and ischemia-reperfusion injury.
Vandria’s proprietary in vitro screening platform facilitates the discovery and optimization of these next-generation mitophagy inducers, enabling the development of targeted therapies for a range of age-related and chronic conditions.
You can view their official pipeline in the below image from their website:
The Company
Vandria was Established in 2021, and is headquartered in Lausanne, Switzerland. The company was spun out of Amazentis SA by founders Patrick Aebischer, Chris Rinsch, and Johan Auwerx. Klaus Dugi is the Chief Executive Officer and he joined Vandria in 2021.
Vandria has successfully secured significant funding to propel its research and development efforts. When emerging from stealth mode in December 2023, Vandria announced a $20.6 million (CHF 18 million) Series A round led by ND Capital, along with a syndicate of high-net-worth private investors.
Recently, in August 2024, the company completed a second closing of its Series A financing, bringing the total raised to $30.7 million (CHF 28.3 million). This round saw participation from Hevolution Foundation and Dolby Family Ventures, joining the lead investor ND Capital.
This funding round was to finance their Phase 1 trial of VNA-318, and to plan for the following efficacy-focused Phase 1b / 2a trials with the first expected to initiate by Q1 of 2026 or potentially earlier.
Vandria is looking to raise a Series B round later in 2025 in order to fund their planned Phase 2 trials and to progress the other pipeline programs.
If you want to learn more about Vandria you can view their official website through this link.
Read about more anti-aging companies on this website through this link.




